
CABA-HFPEF-DZHK27
CAtheter-Based ablation of Atrial fibrillation vs. conventional treatment in patients with Heart Failure with Preserved Ejection Fraction (CABA-HFPEF)
Sponsor´s Protocol Code: CABA-HFPEF-DZHK27
ClinicalTrials.gov Identifier: NCT05508256
STUDY DESIGN
Investigator-initiated multicentre study with prospective parallel group comparison with randomised, open group allocation and blinded endpoint analysis (so-called PROBE design)
Study objective
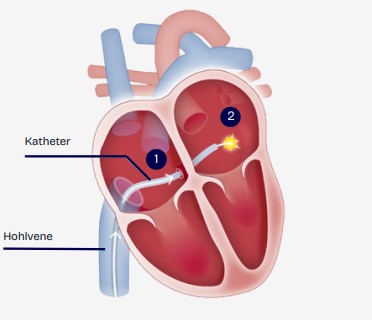
The aim of the study is to investigate whether catheter ablation of atrial fibrillation (AF) can prevent serious cardiovascular events in patients with heart failure with preserved or mildly reduced ejection fraction (HFpEF or HFmrEF).
Description
Atrial fibrillation is an irregular heart rhythm in which the upper chambers of the heart (atria) beat very quickly. Atrial fibrillation can increase the risk of blood clots forming in the heart. These can break loose and travel to the brain, where they can trigger a stroke. Catheter ablation is a procedure that restores the normal heart rhythm. However, even patients who successfully undergo this treatment may have an increased risk of stroke compared to people who have never had atrial fibrillation.
To reduce the risk of stroke, patients are prescribed one of two drugs after catheter ablation: a drug that inhibits the function of blood platelets (acetylsalicylic acid (ASA), e.g. Aspirin®) or a blood thinner (also known as an anticoagulant, e.g. rivaroxaban).The aim of this study is to find out which of these two types of treatment is best at preventing strokes.
CONTACT INFORMATION

PD Dr. med. Abdul Parwani
Principal Investigator
Klinik für Kardiologie, Angiologie und Intensivmedizin
Deutsches Herzzentrum der Charité Berlin